ISO 14155:2011, Clinical investigation of medical devices for human subjects - Good clinical practice: ISO/TC 194, .: Books - Amazon
UNE EN ISO 14155:2012 Clinical investigation of medical devices for human subjects - Good clinical practice (ISO 14155:2011) - European Standards

ISO 14155:2020 CLINICAL INVESTIGATION OF MEDICAL DEVICES FOR HUMAN SUBJECTS UPDATE – BRIDGING THE WAY TO THE EU MDR TRAINING

Guidance on legislation. Clinical investigations of medical devices guidance for manufacturers - PDF Free Download

BS EN ISO 14155:2020 Clinical investigation of medical devices for human subjects. Good clinical practice - European Standards

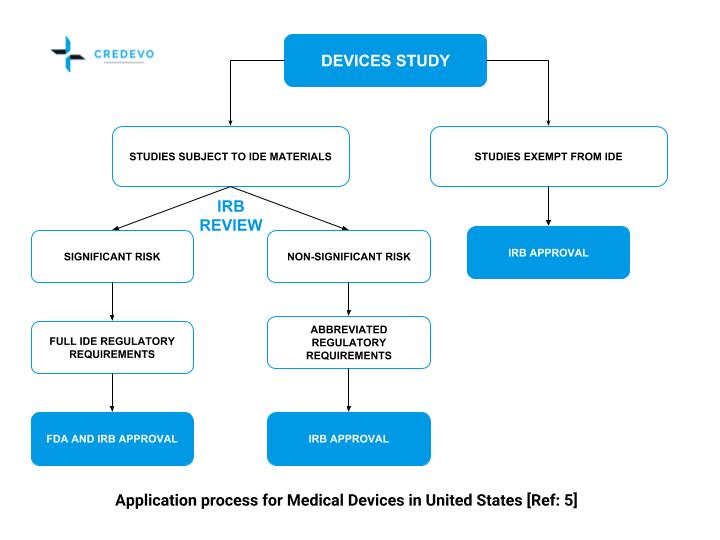
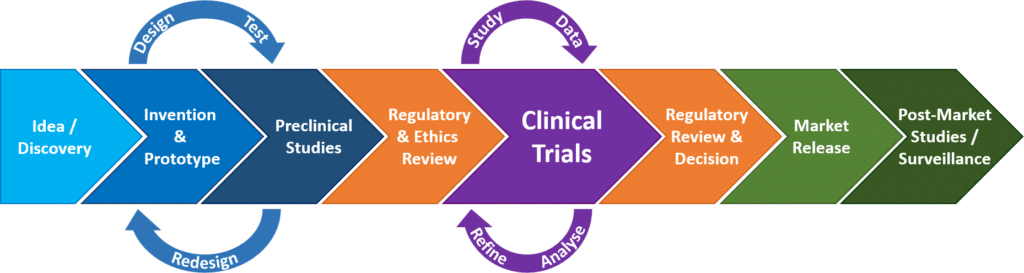
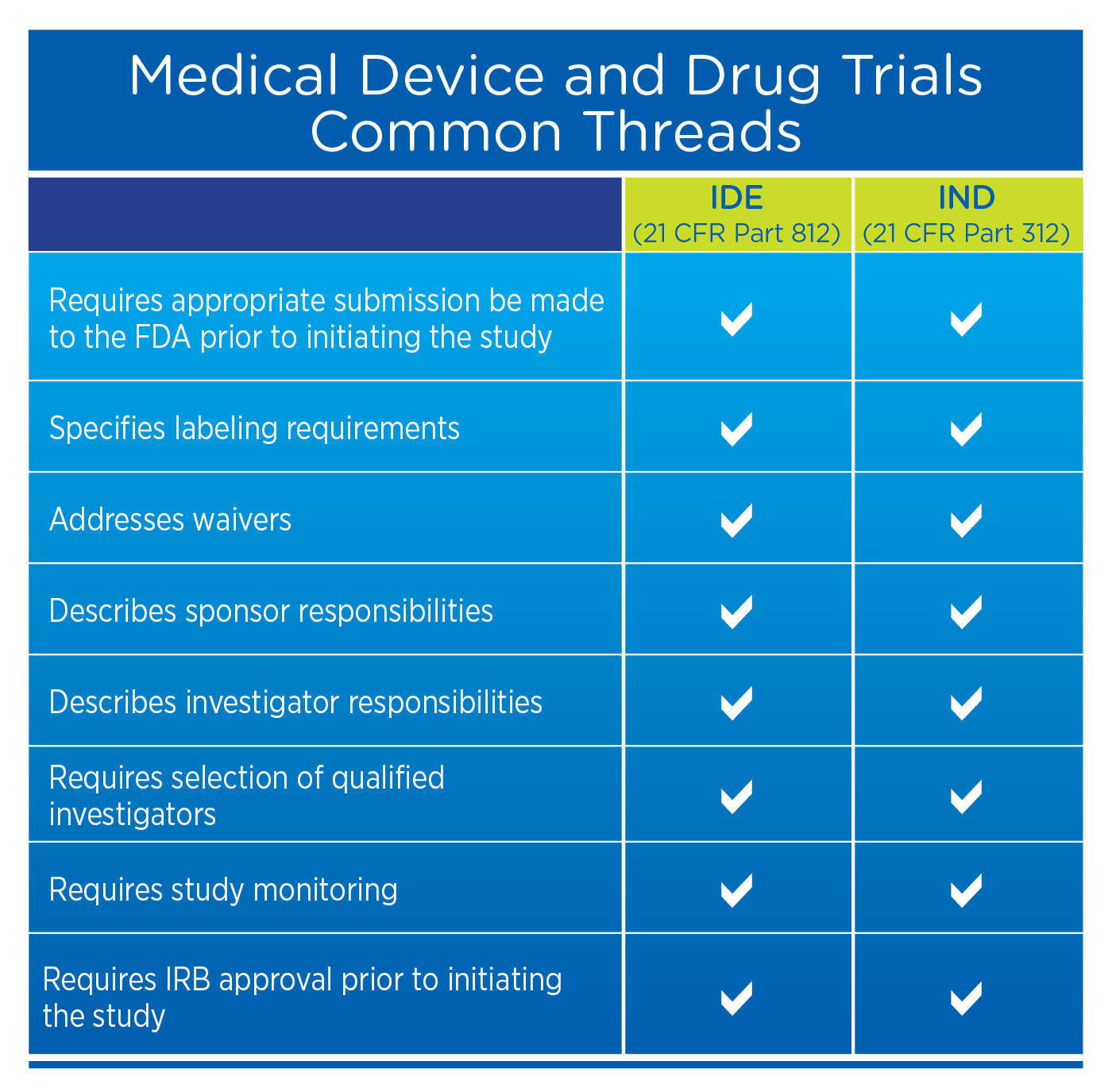
Regulatory aspects of prospective and retrospective clinical research in France in 2018 - ScienceDirect

ANSI/AAMI/ISO 14155-1:2003 - Clinical Investigation Of Medical Devices for Human Subjects - Part 1: General Requirements

ISO 14155-1:2003, Clinical investigation of medical devices for human subjects - Part 1: General requirements: ISO/TC 194: Amazon.com: Books