Oxford Handbook of Clinical and Laboratory Investigation (Oxford Medical Handbooks) - Kindle edition by Provan, Drew. Professional & Technical Kindle eBooks @ Amazon.com.

International Medical Device Clinical Investigations: A Practical Approach: 9781574910544: Medicine & Health Science Books @ Amazon.com

Understand Medical Investigations - Global Healthcare Guide, Magazine and Consultancy by Dr Prem Jagyasi

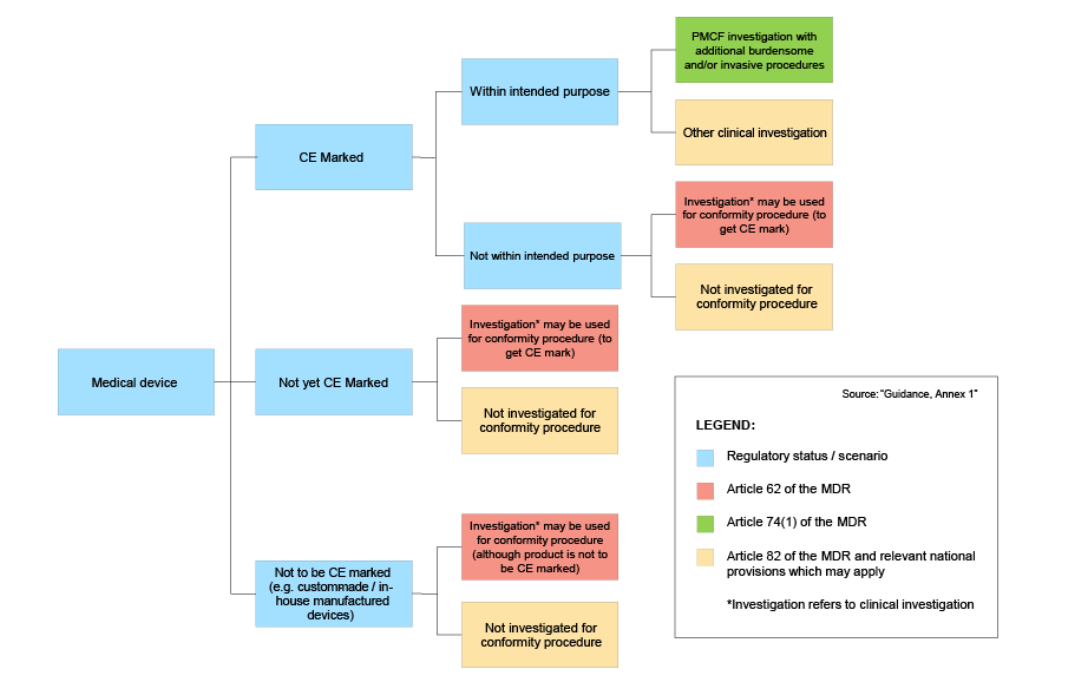
Improved clinical investigation and evaluation of high-risk medical devices: the rationale and objectives of CORE–MD (Coordinating Research and Evidence for Medical Devices) in: EFORT Open Reviews Volume 6 Issue 10 (2021)